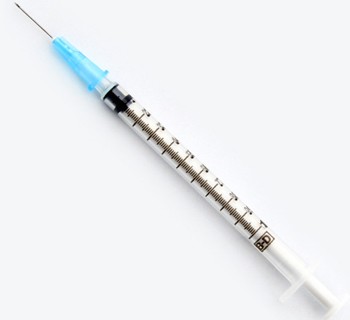
309626 – 1 mL BD slip-tip syringe with 25 G x 5/8 in. BD Tuberculin Syringe with Detachable Needle, Slip Tip. (100/sp, 800/ca).
Our luer slip tip provides a friction-fit connection that requires you to push and twist the syringe tip into the needle hub.
| Detachable Needle | Product has a Detachable Needle |
| Hub Color | Blue |
| Hub Material | Polypropylene |
| Hub Type | Luer |
| Needle Gauge | 25 G |
| Needle Gauge (m) | 0.50 mm |
| Needle Length (in.) | 5/8 in. |
| Needle Length (m) | 15.88 mm |
| Needle Tip Type | Regular |
| Needle Type | Hypodermic |
| Needle Wall Type | Regular |
| Priming Volume | 0.086 mL |
| Pyrogen Free | Product is pyrogen free |
| Syringe Tip Orientation | Concentric |
| Syringe Tip Type | Slip tip |
| Syringe Scale | 0.01 mL graduations |
| Total Shelf Life | 1825 |
| Sterile | Sterilized product |
| Volumetric Accuracy | +/- 5% *for 1ml or smaller,accuracy below 0.2ml is+/- 0.07ml |
| Sterilization Method | EO, Radiation |
| BPA Free | Not made with BPA |
| DEHP Free | Not made with DEHP |
| Latex Statement | Not made with natural rubber latex |
| PVC Free | Not made with PVC |
| Disposable | Disposable product |
| Single Use | Product is for single use only |
BD Products comply with the regulatory requirements of the region in which these are sold and manufactured.
All products which are labeled as #sterile# and released for sale by BD are certified to be sterile as long as the package is unopened and undamaged. For those products labeled #sterile fluid path#, only the fluid path is sterile.
This product is primarily sterilized via E-beam. Sterilization cycle development/validation is performed to 10-6 SAL in accordance with current ISO 11137 guidelines.
This product has been evaluated in accordance with ISO 10993 “Biological Evaluation of Medical Devices”, and complies with all relevant sections.
All products which are labeled as non-pyrogenic and released for sale by BD have been tested per United States Pharmacopeia (USP) chapter 85 – Bacterial Endotoxins Test and meets limits as specified in chapter 161- Transfusion and Infusion Assemblies and Similar Medical Devices.
Representative production samples are collected and inspected in accordance with current applicable product specifications. Inspection records are reviewed and signed off by qualified personnel for product release. The released devices meet applicable BD product specifications.
| What MRI safety information does the labeling contain? | Labeling does not contain MRI Safety Information |
| Device required to be labeled as containing natural rubber latex or dry natural rubber (21 CFR 801.437) | No |
| Device labeled as “Not made with natural rubber latex” | No |
| For Single-Use | Yes |
| Prescription Use (Rx) | Yes |
| Over the Counter (OTC) | No |
| Kit | No |
| Combination Product | No |
| Human Cell, Tissue or Cellular or Tissue-Based Product (HCT/P) | No |
| Packaging Level | Shelfpack | Case | Each |
| Quantity | 100 | 800 | 1 |
| Length | 210.0 mm | 465.0 mm | |
| Width | 178.0 mm | 295.0 mm | |
| Height | 154.0 mm | 285.0 mm | |
| Weight | 850.0 g | 6.8 kg | 8.5 g |
At Health Supply 770 Inc. speed is key. We take great pride in our ability to quickly prepare quotes, ship, and deliver high-quality medical equipment . Which makes us the ideal destination for all your medical needs.

Customer Service: 770-874-0431
[email protected]
7111 Bayou George Dr. Panama City
Florida 32404