
BD Veo insulin syringes feature the shortest BD Ultra-Fine needle, 53% shorter than the 12.7-mm needle. This length is supported by recent recommendations published in Mayo Clinic Proceedings that advocate using the shortest needle first-line for all patient categories. Preferred by most patients over their current needle length, BD Veo insulin syringes provide approximately 8x-lower risk of intramuscular injection compared to a 12.7-mm needle.
Longer needle lengths may lead to frequent IM vs subcutaneous tissue injections, which may cause unexplained hypoglycemia.
Over 80% of patients with diabetes preferred the BD Insulin Syringe with the NEW BD Ultra-Fine 6mm needle to their current needle1,2

| Hub Color | Clear |
| Hub Material | Polypropylene |
| Hub Type | Dart shape |
| Needle Gauge | 31 G |
| Needle Gauge (m) | 0.25 mm |
| Needle Length (in.) | 1/4 in. |
| Needle Length (m) | 6 mm |
| Needle Tip Type | 3-bevel |
| Needle Type | Insulin |
| Needle Wall Type | Thin Wall |
| Syringe Scale | 2 mL graduations |
| Sterile | Sterilized product |
| Sterilization Method | Gamma radiation |
| Latex Statement | Not made with natural rubber latex |
| Disposable | Disposable product |
Most people prefer short needles. They are more comfortable and less threatening, so its easier to start injecting insulin. Short needles also help prevent injections from going into muscle, which can result in pain and poor blood sugar control.
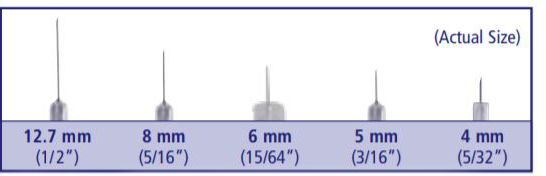
Insulin needles come in different lengths. BD Original Needles are 12.7 mm (1/2″) long, and BD Short Needles are 8 mm (5/16″) long. The BD Nano Needle 4mm (3/16″) and BD Mini Needle 5mm (5/32″) are available only in pen needles. For a comfortable injection experience, shorter needle lengths are recommended and preferred.
The higher the gauge, the thinner the needle. For example, 32G is thinner than a 31G needle.Insulin needles are available in many gauges (G), or thicknesses. The higher the gauge, the thinner the needle. For example, a 32 G needle is actually thinner and more comfortable than a 29 G needle.
Thin Wall: As shown in the diagram, the thin wall needle has a narrower steel wall, allowing a greater volume of fluid to pass through it. The flow rate is typically equivalent to that of a needle one gauge larger. This is especially important with very thin needles.

Most commonly found in insulin and ‘tuberculin’ syringes. Permanently attached needles, also known as integral needles, reduce the amount of medication waste and allow accurate mixing of different medications into one syringe.
BD Products comply with the regulatory requirements of the region in which these are sold and manufactured.
All products which are labeled as #sterile# and released for sale by BD are certified to be sterile per EN 556-1 Sterilization of Medical Devices as long as the package is unopened and undamaged. This product is sterilized via Cobalt 60 – Irradiation. Sterilization cycle development/validation is performed to 10-6 SAL in accordance with current ISO 11137 guidelines.
This product has been evaluated in accordance with ISO 10993 “Biological Evaluation of Medical Devices”, and complies with all relevant sections.
All products which are labeled as non-pyrogenic and released for sale by BD have been tested per United States Pharmacopeia (USP) chapter (85)- Bacterial Endotoxins Test and meets limits as specified in chapter 161- Transfusion and Infusion Assemblies and Similar Medical Devices.
Representative production samples are collected and inspected in accordance with current applicable product specifications. Inspection records are reviewed and signed off by qualified personnel for product release. The released devices meet applicable BD product specification(s).
| What MRI safety information does the labeling contain? | Labeling does not contain MRI Safety Information |
| Device required to be labeled as containing natural rubber latex or dry natural rubber (21 CFR 801.437) | No |
| Device labeled as “Not made with natural rubber latex” | No |
| For Single-Use | Yes |
| Prescription Use (Rx) | No |
| Over the Counter (OTC) | No |
| Kit | No |
| Combination Product | No |
| Human Cell, Tissue or Cellular or Tissue-Based Product (HCT/P) | No |
Proper injection technique is essential to improve consistency in medication delivery and optimize glycemic control. It includes factors such as:
Proper syringe disposal will help to:
| Packaging Level | Shelfpack | Case | Each |
| Quantity | 100 | 500 | 1 |
| Length | 17.145 cm | 43.82 cm | |
| Width | 13.97 cm | 18.42 cm | |
| Height | 8.415 cm | 15.24 cm | |
| Weight | 390.0 g | 2.15 kg | 4.545 g |
At Health Supply 770 Inc. speed is key. We take great pride in our ability to quickly prepare quotes, ship, and deliver high-quality medical equipment . Which makes us the ideal destination for all your medical needs.

Customer Service: 770-874-0431
[email protected]
7111 Bayou George Dr. Panama City
Florida 32404