In a time when people are becoming more aware of the uniqueness of their bodies and the slogan of ‘all body types fit’ can be frequently heard, there is a group that is rushing towards any medicated option to reduce weight. This is the reason many weight loss drugs are flying off the shelves so quickly. Among these weight-reducing drugs, tirzepatide, marketed under the brand name of Mounjaro, is a new addition. Eli Lilly and Co. Ltd., the US-based pharma company, carries the patent rights for its production and marketing. It has been claimed that, with the regular use of tirzepatide, up to 16% of body weight can be lost in a short amount of time. Let us take a look at what Mounjaro actually is and how much is it better as compared to Ozempic and Wegovy, the commonly used weight loss drugs.
Trizepatide-What is it?
Tirzepatide, in fancy scientific terms, is a GLP-1 agonist which works by mimicking the role of an endogenous hormone that aids in reducing the appetite and subsequently the food intake. The drug has been approved by the FDA in 2022 for the treatment of type II diabetes. However, its parent company is expecting to get another FDA approval regarding its use as a weight loss agent as soon as the end of this year.

Mounjaro (Tirzepatide) subcutaneous injection by Eli Lilly & Co.
How does tirzepatide work?
Mounjaro is to be administered once weekly as a subcutaneous injection for the purpose of losing weight. The drug works by slowing the gastric emptying time i.e. once a person consumes food, it stays in the stomach for a longer period making the individual feel fuller for longer.
Studies regarding the efficacy of tirzepatide as a weight loss drug
A phase 3 double-blind randomized investigation, published in the New England Journal of Medicine (NEJM), has been conducted to evaluate the efficacy of tirzepatide in weight loss. More than 2500 diabetes-free individuals with weight-related complications were recruited for the study who were further divided into groups. Each group received a different subcutaneous dose of the test drug tirzepatide i.e. 5 mg, 10 mg, and 15 mg respectively. The drug was administered once weekly against a placebo, the tablet without any tirzepatide given for comparison, for a period of 72 weeks. The results of the study indicated a strong relationship between the amount of drug used and the percentage of weight lost in the specified period of timeas the groups which received different concentrations of tirzepatide lost up to 15.7% of their body weight in comparison to those in the control group who lost 3.3% of it. This manifests the weight loss claims about the effectiveness of tirzepatide to be true.
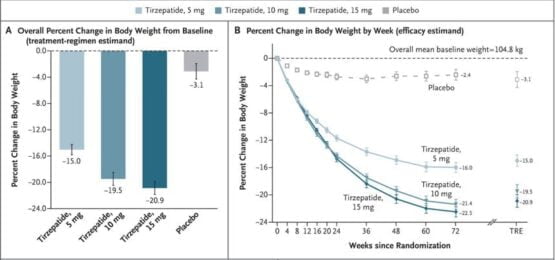
Graphs showing the percentage weight loss after the use of different concentrations of tirzepatide
Studies, similar to this one, have been conducted in various countries to obtain evidence of the effectiveness of tirzepatide in genetically distinct populations. These investigations will serve as a base for the FDA approval needed for the remarketing of Mounjaro as an efficient weight loss drug.
Is tirzepatide better than semaglutide?
Ozempic and Wegovy, both are FDA-approved weight loss drugs that carry an active ingredient called ‘Semaglutide’. Their efficacy has been proven by multiple studies which claim a weight loss falling in the range of 6.2 to 12.4% with a 68-week dosing. Although this value is prominent as compared to the one achieved by the placebo, the Mounjaro (tirzepatide) has won the race by increasing the percentage weight loss up to 16% thus earning the title of ‘King Kong’ drug.
Comparison of Ozempic and Wegovy (Semagluitide) with Mounjaro (Tirzepatide)
| Brands | API* | Dosage | Route of administration | FDA approval for | Intended age group | Additional benefits |
| Ozempic (Novo Nordisk) | Semaglutide | Once weekly | Intravenous | Type II diabetes | Adults | Weight loss |
| Wegovy (Novo Nordisk) | Semaglutide | Once weekly | Intravenous | Weight loss | Kids > 12 years of age, adults | – |
| Mounjaro (Eli Lilly) | Tirzepatide | Once weekly | Intravenous | Type II diabetes | Adults | Weight loss |
*API=Active pharmaceutical ingredient
Side effects of tirzepatide
Like the other weight loss drugs, the use of tirzepatide is also not free from side effects. However, these are minor in nature, they may become significant in some patients. These include:
- Nausea
- Diarrhea
- Decreased appetite
- Vomiting
- Constipation
- Indigestion
- Abdominal pain
Moreover, when a comparison was made at the level of side effects, the impacts of semaglutide were more pronounced as compared to those experienced with different concentrations of tirzepatide. This characteristic has rendered the latter superior to the prior drug.
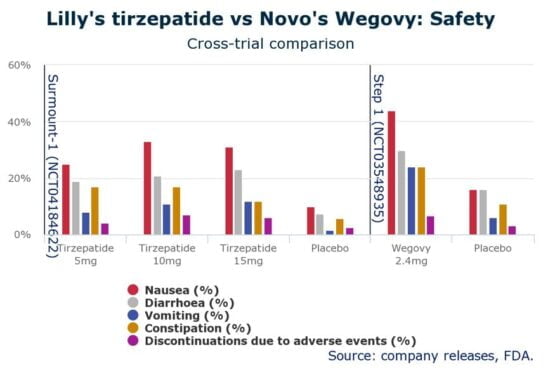
Comparison of side effects of tirzepatide and semaglutide
Conclusion
Society, nowadays, has become extremely health-conscious in many ways. Almost every day, a celebrity or a social media influencer can be seen promoting exercising, eating more greens, using collagen supplements, adding bone broth into your diet, or simply preferring water over other drinks. Keeping this in mind, it is safe to say that health has become a top priority for people of this age and time. One of these matters of extreme importance is maintaining a healthy weight and getting rid of any extra pounds. Another way to say this is that obesity is not in fashion anymore. Semaglutide and tirzepatide are the pharmaceutical moieties that are being used for weight loss in recent times. When compared at different levels with semaglutide, tirzepatide has been proven superior in its effects hence it is an emerging weight loss agent in the coming years.

PhD Scholar (Pharmaceutics), MPhil (Pharmaceutics), Pharm D, B. Sc.
Uzma Zafar is a dedicated and highly motivated pharmaceutical professional currently pursuing her PhD in Pharmaceutics at the Punjab University College of Pharmacy, University of the Punjab. With a comprehensive academic and research background, Uzma has consistently excelled in her studies, securing first division throughout her educational journey.
Uzma’s passion for the pharmaceutical field is evident from her active engagement during her Doctor of Pharmacy (Pharm.D) program, where she not only mastered industrial techniques and clinical case studies but also delved into marketing strategies and management skills.
Throughout her career, Uzma has actively contributed to the pharmaceutical sciences, with specific research on suspension formulation and Hepatitis C risk factors and side effects. Additionally, Uzma has lent her expertise to review and fact-check articles for the Health Supply 770 blog, ensuring the accuracy and reliability of the information presented.
As she continues her PhD, expected to complete in 2025, Uzma is eager to contribute further to the field by combining her deep knowledge of pharmaceutics with real-world applications to meet global professional standards and challenges.








